What is Hemoglobin:
Hemoglobin is a protein in your red blood cells that carries oxygen to your body’s organs and tissues and transports carbon dioxide from your organs and tissues back to your lungs.Hemoglobin (Hb) is the most studied of the heme containing globulin proteins and yet is not fully understood. It was one of the first proteins to be studied by X-ray crystallography, and earned Max Perutz the Nobel Prize in Chemistry in 1962.
Function of hemoglobin:


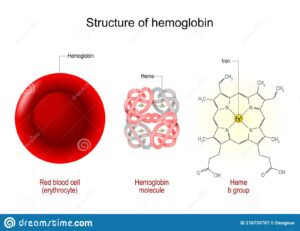
Structure of Hemoglobin:
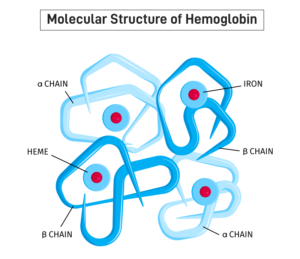
Hemoglobin comprises four subunits, each having one polypeptide chain and one heme group . All hemoglobins carry the same prosthetic heme group iron protoporphyrin IX associated with a polypeptide chain of 141 (alpha) and 146 (beta) amino acid residues. The ferrous ion of the heme is linked to the N of a histidine. The porphyrin ring is wedged into its pocket by a phenylalanine of its polypeptide chain. The polypeptide chains of adult hemoglobin themselves are of two kinds, known as alpha and beta chains, similar in length but differing in amino acid sequence. The alpha chain of all human hemoglobins, embryonic and adult, is the same.
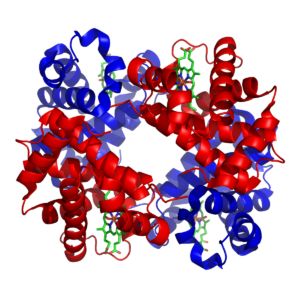
The structure of hemoglobin has been extensively studied by x-ray analysis (6). The arrangement of the subunits—which is known as the quaternary structure—differs in the oxy- and deoxyhemoglobin.
In human hemoglobin, the fit between the polypeptide chain is critical because the gap between two of the polypeptide chains in the hemoglobin molecule becomes narrower when oxygen molecules become attached to the ferrous atoms. This has been likened by Max Perutz to a molecular form of paradoxical breathing: unlike the lungs, the hemoglobin molecule contracts when oxygen enters and expands when oxygen leaves.